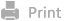Engineered Bacterial Outer Membrane Vesicles-Based Doxorubicin and CD47-siRNA Co-Delivery Nanoplatform Overcomes Immune Resistance to Potentiate the Immunotherapy of Glioblastoma
Time:2025/3/5 14:19:00 Views:2631
Apart from the blood-brain barrier (BBB), the efficacy of immunotherapy for glioblastoma (GBM) is limited by the presence of intrinsic and adaptive immune resistance, implying that co-delivery of various immunotherapeutic agents or simultaneous regulation of different cells is urgently needed. Bacterial outer membrane vesicles (OMVs) offer a unique advantage in the treatment of GBM, owing to their multifunctional properties as carriers and immune adjuvants and their ability to cross the BBB. However, traditional OMVs can lead to toxic side effects and disruption of tight junctions in the BBB. Therefore, to enhance the in vivo safety and targeting capability of OMVs, we introduced engineered OMVs to reduce toxicity and further constructed a modularly assembled nanoplatform by performing simple peptide modifications. This nanoplatform demonstrates satisfactory biosafety and is able to continuously cross the BBB and target GBM with the assistance of Angiopep-2. Subsequently, immunogenic substances on OMVs, along with carried small-interfering RNA (siRNA) and doxorubicin, can promote and enhance the reprogramming and phagocytic abilities of glioma-associated microglia and macrophages (GAMs), respectively, and increase the immunogenicity of GBM, ultimately overcoming GBM immune resistance to enhance the efficacy of immunotherapy. This OMVs-based nanoplatform provides a new paradigm and insights into the development of immunotherapy for GBM. The related research results were published online in the international renowned journal Advanced Materials (IF=27.4) under the title “Engineered Bacterial Outer Membrane Vesicles-Based Doxorubicin and CD47-siRNA Co-Delivery Nanoplatform Overcomes Immune Resistance to Potentiate the Immunotherapy of Glioblastoma”.
Current immunotherapies for GBM have not shown significant survival benefits in patients. In addition to the blocking effect of the BBB, GBM is also classified as a highly “cold” tumor, characterized by “high intrinsic immune resistance” with extremely low immunogenicity and “high adaptive immune resistance” with overexpression of multiple checkpoint ligands and extensive infiltration of immunosuppressive cells in the tumor microenvironment (TME). Among them, CD47 is a classic phagocytic checkpoint protein that can prevent GAMs from phagocytosing GBM cells. In contrast, GAMs have the highest proportion among all immune cells and mainly exist in a pro-tumor phenotype. Studies on the immune resistance mechanism of GBM have shown that the synergistically overcoming both of intrinsic and adaptive immune resistance is crucial for improving the efficacy of immunotherapy, which means that multiple immunotherapeutic agents need to be delivered simultaneously or different cells need to be regulated simultaneously. However, the differences in physicochemical properties of different immunotherapeutic agents pose considerable challenges for the selection of carriers and the design of suitable co-delivery systems.
OMVs are nanoscale bilayer lipid vesicles produced spontaneously by Gram-negative bacteria. Similar to exosomes, OMVs serve as natural carriers capable of efficiently loading various agents, including small-molecule hydrophobic chemotherapeutics and large-molecule genetic agents, while ensuring their stability and prolonged circulation in vivo. Additionally, OMVs inherit numerous immunogenic components from the outer membrane of the parent bacteria and periplasm, which makes them potential vaccines or immune adjuvants to stimulate the immune system and exert antitumor effects. Notably, no study has investigated the potential of OMVs in enhancing immune responses against GBM. More importantly, OMVs can cross the BBB through various mechanisms (e.g., the interaction between outer membrane proteins and gp96 and hitchhiking on neutrophils), offering a unique natural advantage in treating GBM. However, it is worth noting that, whereas OMVs exhibit reduced toxicity compared to bacteria, attention must still be given to the triggering of cytokine storms by the large amounts of toxic substances they carry. Moreover, although OMVs can increase BBB permeability to some extent under the action of lipopolysaccharide (LPS), this is based on the disruption of tight junctions within the BBB. In fact, this kind of disruption is not what we desire and may even require certain measures to prevent its occurrence.
To improve the in vivo safety of OMVs, we employed genetic engineering to knock out the lipid A acyltransferase gene (msbB) of Escherichia coli (E. coli) BL21 to produce attenuated OMVs. Building on this, we developed a modular nanoplatform, Angiopep-2-OMVs@PEG3.5k-TK-PEI1.8k/siCd47&aptamer-DOX (AO@PTP/47aD), for the co-delivery of siCd47 and DOX to comprehensively disrupt immunes resistance in GBM. The core of this nanoplatform was composed of a cationic polymer complexed with both siCd47 and a DOX-loaded aptamer through electrostatic interactions, whereas the outer layer consisted of ΔmsbB OMVs. Additionally, Angiopep-2 was incorporated onto the surface of ΔmsbB OMVs via simple lipid fusion to enhance targeting specificity. We demonstrated that AO@PTP/47aD exhibited excellent biosafety and considerable ability to cross the BBB and target GBM. Under the action of high concentrations of reactive oxygen species (ROS) and adenosine triphosphate (ATP) in GBM cells, DOX and siCd47 are rapidly released, and DOX overcomes the intrinsic immune resistance of GBM by triggering ICD, subsequently promoting the maturation of dendritic cells (DCs) and activation of T cells. On the other hand, certain immunogenic substances in ΔmsbB OMVs and the released siCd47 could initiate the reprogramming of GAMs and knock down the CD47 of GBM cells respectively to overcome adaptive immune resistance, which enhanced the antitumor effect (such as the release of pro-inflammatory cytokines) and phagocytosis of GAMs toward GBM cells. Ultimately, this OMVs-based co-delivery system elicited a robust and effective antitumor immune response through a dual-pronged strategy that considerably inhibited tumor growth in an orthotopic GBM mouse model.

Scheme 1. The modular assembly of nanoplatform (AO@PTP/47aD) and the mechanism of boosting the immunotherapy in GBM through a dual-pronged strategy of overcoming the intrinsic and adaptive immune resistance.
Haoyu You, the PhD candidate from the School of Pharmacy, Fudan University, is the first author. Associate Professor Tao Sun and Professor Chen Jiang are the corresponding authors of this paper. The work was supported by grants from the National Natural Science Foundation of China, National Key R&D Program of China, Shanghai Municipal Science and Technology Major Project, and ZJLab.
For more information:https://advanced.onlinelibrary.wiley.com/doi/full/10.1002/adma.202418053