Transcytosis Mediated Deep Tumor Penetration for Enhanced Chemotherapy and Immune Activation of Pancreatic Cancer
Recently,
our group has developed a versatile drug delivery system that can trigger
transcytosis. The delivery system can efficiently deliver chemotherapy drugs to
the deep tumor, regulate the microenvironment, and activate the anti-PDAC
immune response. The study was published online in Advanced Functional
Materials, named Transcytosis Mediated Deep Tumor Penetration for
Enhanced Chemotherapy and Immune Activation of Pancreatic Cancer.
The
hyperproliferative tumor stroma of pancreatic ductal adenocarcinoma (PDAC)
severely limits drug permeation and constructs an immunosuppressive
microenvironment, causing resistance to chemotherapy and immunotherapy.
Traditional nanomedicine mainly focuses on manipulating nanoparticles’ particle
size or electrical characteristics to penetrate deep PDAC through the
paracellular pathway, but the transcellular pathway is often ignored. Transcytosis
has become an important way of transporting macromolecular nutrients in tumors,
as the conventional transport routes cannot sustain their exuberant cellular
metabolic demands. Therefore, the rational design of nanomedicines that can
trigger transcytosis would provide a rational strategy to bypass the long-term
bottleneck of nanomedicine delivery in anti-PDAC therapy.
As
a typical cold tumor, PDAC confronts inadequate immune cell activation and
infiltration, which limit the immune response against PDAC. The stimulator of
interferon genes (STING) pathway has arisen as a promising target for
energizing anti-tumor immune responses. It simultaneously provides new
alternations for immunotherapeutic strategies in PDAC treatment. A lately
reported polymer with cyclic tertiary amine structures (PHMI) exhibited
STING-dependent immune activation ability. Meanwhile, as the primary driver of
desmoplasia in PDAC, FAK acts as a critical molecular fulcrum in reshaping the
tumor microenvironment and affecting the infiltration of both drugs and immune
cells into the deep tumor. Other than STING activation, PHMI can also sense the
mild acidity in tumors, and realize the hydrophobicity to hydrophilicity
transformation. This feature energizes PHMI as an advanced FAK inhibitor
delivery carrier and anti-tumor immune response stimulator synergistically.

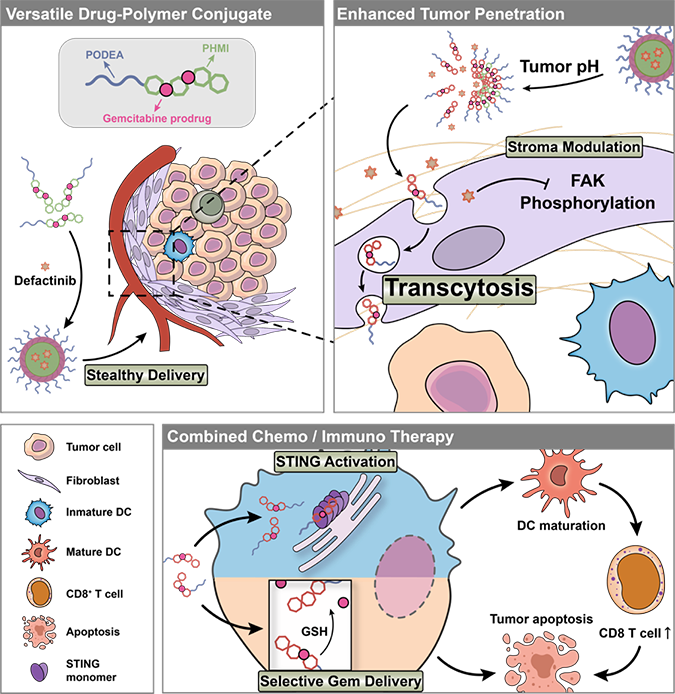
Herein,
a multifunctional drug-polymer conjugate PODEA-Gem-HMI which can trigger
transcytosis, was designed, prepared, and assembled into dual-responsive
nanoparticles for the codelivery of chemotherapeutic drug gemcitabine and
selective FAK inhibitor defactinib. PODEA is a zwitterion polymer showing high
phospholipid affinity but not to proteins, which guarantees prolonged
systematic circulation. Upon reaching the tumor tissue, the nanoparticle would
disintegrate and release defactinib in the TME as sensing the specific mild
acidity. And the unique structure of PODEA enables the reversible binding
between the conjugates and cell membranes and induces adsorption-mediated
transcytosis, promoting tumor extravasation and penetration. To avoid
gemcitabine being taken up by non-tumor cells and causing unwanted side
effects, we conjugated gemcitabine to the polymer backbone with a
reduction-responsive disulfide bond. When the conjugate is internalized by
tumor cells, the overexpressed intracellular GSH can destroy the disulfide
bonds and release gemcitabine to restrain tumor proliferation. Meanwhile, after
cellular uptake, PHMI would activate the STING pathway and promote cytokine
expression.
In
short, this versatile drug-polymer conjugate constituted multifunctional
nanoparticle can prerelease defactinib triggered by mild acidity in the TME,
transport gemcitabine to the deep tumor through transcytosis, and activate the
STING pathway for TME modulation, chemotherapy promotion, and spontaneous
immune activation, providing a promising combined strategy for PDAC treatment.
Hongyi
Chen, the PhD fellow from the School of Pharmacy, Fudan University, is the
first author. Professor Chen Jiang is the corresponding author, and Associate
Professor Sun Tao is the co-corresponding author of this paper. The work was
supported by grants from the National Natural Science Foundation of China, Key
Projects of Shanghai Science Foundation, Shanghai Municipal Science and
Technology Major Project, and ZJLab.
For
more information: https://doi.org/10.1002/adfm.202214937