Bioinspired nanoerythrocytes for metabolic microenvironment remodeling and long-term prognosis promoting of acute ischemic stroke

Recently, our group has developed a
nanoerythrocyte, which could regulae the metabolic microenvironment of the
brain lesion area and was applied for the whole process of acute ischemic
stroke (AIS) treatment. Recently, related article was published online in the
internationally renowned journal Nano Today (IF=18.9) under the title of “Bioinspired
nanoerythrocytes for metabolic microenvironment remodeling and long-term
prognosis promoting of acute ischemic stroke”.
In recent years, the importance of the complex
microenvironment of brain in the treatment of brain diseases has become
increasingly prominent. Its basic unit is the neurovascular unit, which extends
outward from the brain capillary endothelial cells that constitute the BBB,
including pericytes, glial cells, nerve cells, and extracellular matrix. The
mutual coupling and normal metabolic activities between cells are the basic
elements to maintain the homeostasis of the microenvironment. Cell metabolites
are important components of the microenvironment, including cytokines, gas
molecules, and nutrients. The levels of these metabolites in turn influence the
fate of the cell. Therefore, the metabolic homeostasis of cells in the
microenvironment of the brain is critical to the overall microenvironment.
In AIS, the characteristics of the metabolic
microenvironment mainly include the following two aspects. The first is
abnormalities in neuronal metabolism, manifested by a marked downregulation of
cellular oxygen, glucose and energy metabolism. This is followed by metabolic
abnormalities in other cells in the microenvironment, such as glial cells and
endothelial cells. Significant upregulation of reactive oxygen species,
pro-inflammatory factors, and osmolarity was manifested. Metabolic
abnormalities of other cells in the microenvironment will put metabolic stress
on neurons, which will aggravate disease damage. Studies have shown that there
is a unique internal mechanism of metabolic microenvironment damage in stroke.
In the stage of hypoxia-ischemia, HIF-1α enters the nucleus in response to the
hypoxic microenvironment, activates the expression of VEGF, and then causes the
destruction of the BBB and the damage of microcirculation. In the later stage
of reperfusion, with the excessive oxygen level, a large amount of ROS is
produced, causing mitochondrial damage and ATP generation blockage. In
addition, Akt/GSK-3β, a key signaling molecule in glucose metabolism, is
dysregulated, leading to a decrease in glucose uptake and glycolysis. In
summary, we hope to provide a basis for the design of the drug delivery system
by understanding the changes in the metabolic microenvironment of AIS, so as to
treat the disease in a targeted manner.
Therefore, we designed our drug delivery system
based on the following three key changes in the metabolic microenvironment
(Figure 1). First of all, the BBB function is impaired in AIS, which is
manifested by the presence of microthrombus deposition at the vascular injury,
which suggests that we can use thrombus-binding peptides as the targeting head
group of the drug delivery system to increase the residence time of the lesion
in the brain. Secondly, the contradiction of oxygen metabolism in the metabolic
microenvironment is mainly manifested in the double-edged sword role played by
oxygen before and after reperfusion, and the hemoglobin in the organism is a
natural oxygen regulator, and as a carrier of the drug delivery system, it can
Binds or releases oxygen reversibly. Furthermore, the level of glucose
metabolism in neurons is significantly inhibited, and the model drug
pyrroloquinoline quinone can activate the Akt/GSK-3β signaling pathway in
cells, thereby correcting abnormal glucose metabolism in cells.
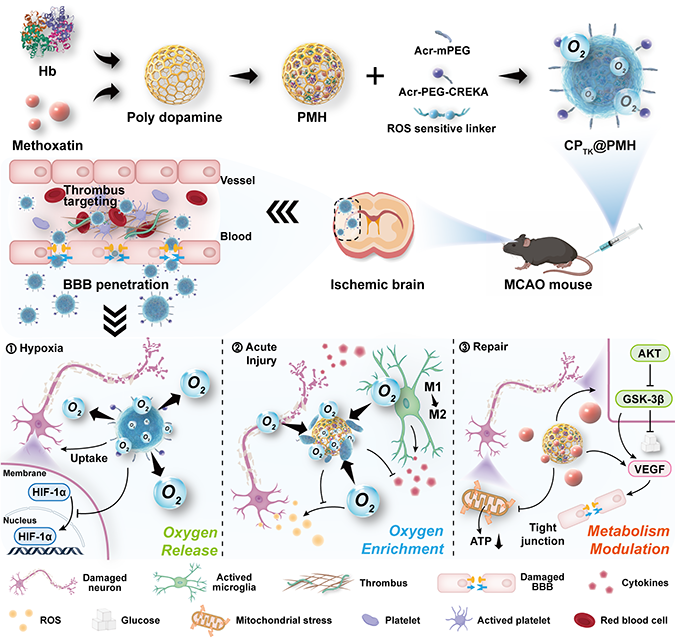
Figure 1. Schematic diagram of the construction of nanoerythrocytes and the
modulation of the metabolic microenvironment in the whole process of AIS.
By targeting micro-thrombus, nanoerythrocytes could
actively reach the ischemic core and enhance drug accumulation. During the
ischemic phase, the nanoerythrocytes could release oxygen in response to the
hypoxic microenvironment to relieve hypoxia. During the reperfusion phase,
nanoerythrocytes could combine with excess oxygen to block the generation of
ROS and promote the polarization of microglia, finally inhibiting the acute
injury during the reperfusion phase. In the late recovery stage,
nanoerythrocytes released drugs to regulate the Akt/GSK-3β signaling pathway to
activate glucose metabolism and achieved BBB protection. During the progression
of AIS, key metabolic elements, including oxygen balance and glucose
metabolism, were positively regulated, and satisfactory outcomes were obtained
in pMCAO (permanent embolization model) and tMCAO (short-term embolization
model) mouse models, respectively. Short-term and long-term therapeutic
effects. Therefore, this work contributes to the understanding of the
pathological process in different stages of AIS and suggests that metabolic
microenvironment regulation can serve as a potential therapeutic strategy for
AIS. More importantly, compared with recently developed nanomedicines, the
concept of whole-course therapy can realize multi-target and multi-level treatment
of diseases, which may have better clinical application potential.
Peixin Liu, PhD student of 2019 from School of
Pharmacy, Fudan University, is the first author, while Professor Chen Jiang is
the corresponding author. The work was supported by the grants from the National Natural Science Foundation of China (81872808,
82121002), Key Projects of Shanghai Science Foundation (19JC1410800), and
Shanghai Municipal Science and Technology Major Project (Grant 2018SHZDZX01)
and ZJLab.
Link address: https://www.sciencedirect.com/science/article/pii/S1748013223000555#ack0005