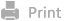ROS-removing nano-medicine for navigating inflammatory microenvironment to enhance anti-epileptic therapy

As a neurological disorder in the brain, epilepsy is not only
associated with abnormal synchronized discharging of neurons, but also
inseparable from non-neuronal elements in the altered microenvironment.
Anti-epileptic drugs (AEDs) merely focusing on neuronal circuits frequently
turn out deficient, which is necessitating comprehensive strategies of
medications to cover over-exciting neurons, activated glial cells, oxidative
stress and chronic inflammation synchronously. Therefore, we would report the
design of a polymeric micelle drug delivery system that was functioned with
brain targeting and cerebral microenvironment modulation. In brief, reactive
oxygen species (ROS)-sensitive phenylboronic ester was conjugated with
poly-ethylene glycol (PEG) to form amphiphilic copolymers. Additionally,
dehydroascorbic acid (DHAA), an analogue of glucose, was applied to target
glucose transporter 1 (GLUT1) and facilitate micelle penetration across the
blood-brain barrier (BBB). A classic hydrophobic AED, lamotrigine (LTG), was
encapsulated in the micelles via self-assembly. When administrated and
transferred across the BBB, ROS-scavenging polymers were expected to integrate
anti-oxidation, anti-inflammation and neuro-electric modulation into one
strategy. Moreover, micelles would alter LTG distribution in vivo with improved
efficacy. Overall, the combined anti-epileptic therapy might provide effective
opinions on how to maximize neuroprotection during early epileptogenesis.
Related research (DOI: 10.1016/j.apsb.2022.09.019) has recently been
available online powered by Acta Pharmaceutica
Sinica B (IF = 14.903). Please kindly refer to https://www.sciencedirect.com/science/article/pii/S2211383522004087 for more information.